

| Prof. Dr. Jürgen Kopf | Research Keywords | |
| PhD (Crystallography/Chemistry): University of Hamburg, 1973 | - X-ray structure analysis | |
| Scientific career: | - Single crystal diffractometry | |
| Postdoc in Berkeley, California, 1978 (Prof. Jolly) | - X-ray structure determination | |
| Habilitation in Hamburg, 1987 | - Computer programming | |
| since 1996 Professor in Hamburg | - Carbohydrates | |
| Memberships: | - Organometallics | |
| GDCh, Gesellschaft Deutscher Chemiker | - Structural Chemistry | |
| DGK, Deutsche Gesellschaft für Kristallographie | ||
| ACS, American Chemical Society | ||
| ACA, American Crystallographic Association | ||
| Phone/Fax: +49-(0)40-42838-2897/6348 | ||
| email: kopf@xray.chemie.uni-hamburg.de | ||
| Internet: http://www.juergenkopf.de/index.html |
a) 1,3-Parallel interactions in alditols (``sugar alcohols'')
Most of the crystal structures of the pentitols and hexitols have been determined at the end of the sixties. From those previous structure determinations it was derived, that the conformation in the crystal is controlled by the ``so-called'' Hassel-Ottar effect: Generally, a planar zigzag conformation in the carbon chain is expected, but in the case when the C(n)-OH and C(n+2)-OH bonds are arranged parallel in the straight-chain conformation, one or more of the C-C-C-C torsion angles adopt the gauche conformation to avoid this unfavorable 1,3-parallel interaction of O atoms (designated as O//O). This claimed general avoidance of O//O interactions (which resembles 1,3-diaxial interactions in the cyclic case) was incorrectly assigned to Hassel and Ottar who, indeed, were very cautious in speculation about steric influence of such a geometry.
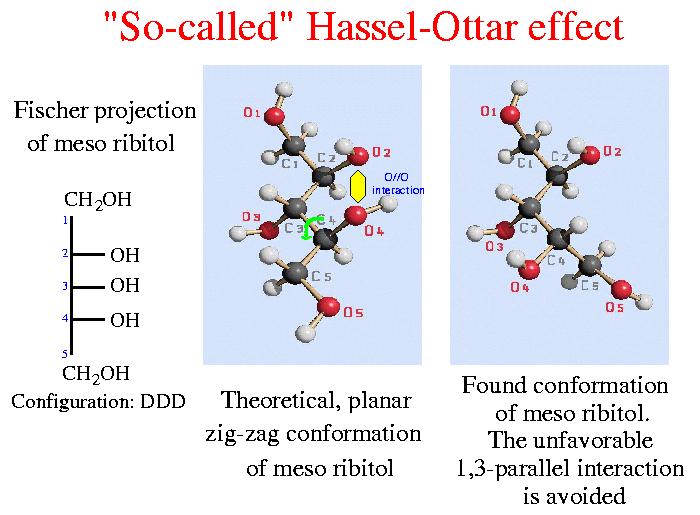
In continuation of our research program we have determined the crystal and molecular structures of previously not treated pentitols, hexitols, heptitols and several higher alditols up to decitols including peracetylated derivatives in most cases. Our investigations show that the general avoidance of 1,3-parallel interactions between the heavy atoms (designated as O//O and C//O, respectively) is not a dominating factor in determinig the conformation of higher alditols in the solid state. Acetylated alditols in particular are able to adopt ``unexpected'' conformations. A systematic search in the Cambridge File [1] for the occurence of such O//O and C//O interactions in the solid state conformations of acyclic carbohydrate derivatives confirms our results. Special patterns of hydrogen bonds can not explain this fact, because acetylated derivatives do not, generally, behave different. The results of our investigations have been summarized [2,3].
b) The application of graphic desktop software in single crystal diffractometry
One of the first scientific instruments to be controlled by a computer was the single crystal diffractometer. Early computer-contolled diffractometers were built at Hilger & Watts (Y290), Enraf-Nonius (CAD4), Philips (PW1000) and Siemens (AED). The programs developed for those instruments were written in assembler, mostly for a DEC PDP-8 computer. The first diffractometer software, completely written in the high-level computer-language FORTRAN IV, was the control program for the Syntex .
The past ten years have seen a revolution in computing and graphics hardware with the arrival of PCs and powerful graphic workstations which become increasingly faster and cheaper. New desktop systems, like GEM, WINDOWS or X/WINDOWS, allow an unexperienced user easy interaction with the computer.

In connection with the electronic rebuilding of a 22 years old, mechanically still reliable Hilger & Watts (Y290) we have developed a new graphically oriented program for the diffractometer control which uses the above mentioned advantages. A new interface, using a 68008-based single-board microcomputer for serving the four stepper motors of the four circles, is connected to an ATARI Mega ST2 via the serial interface RS232. The diffractometer control software is completely written in FORTRAN77 and has the following features:
By isolating all modifications required to drive a different instrument, it was possible in between three weeks to re-write the same control program for an 18 years old, mechanically still reliable SYNTEX diffractometer. This instrument was one of the first diffractometers where the control software was completely written in FORTRAN. The computer used at that time was the (now obsolete) Data General NOVA. Later this computer was replaced by a new ``SIEMENS-interface'' which simulates the Nova 1200. In contrast to the old-fashioned, 2-letter command structure given on a VT100 the new control program uses easy to learn drop-down menus, dialog-, alert- and file-selector-boxes. The availability of the source code allowed the easy implementation of crystal-based psi-angles [4] for numerical absorption corrections with the program ABSPSI [5] and SHELXL97.
Although an ATARI Mega ST2 with 1 Mbyte memory and only 8 MHz processor speed is completely sufficient even to continously display on-line profiles of all scan-types for each reflection, a PC version (Linux/X11) is under development.
c) Funding organisations
Fonds der Chemischen Industrie, (DFG) Deutsche Forschungsgemeinschaft.
d) References
[1] F. H. Allen, O. Kennard, Chemical Design Automation News, 1993, 8, 31
[2] P. Köll, M. Bischoff, C. Bretzke, J. Kopf, Carbohydrat Res., 1994, 262, 1
[3] J. Kopf, M. Morf, B. Hagen, M. Bischoff, P. Köll, Carbohydrate Res., 1994, 262, 9
[4] D. Schwarzenbach, H. D. Flack, J. Appl. Cryst., 1989, 22, 601
[5] N. W. Alcock, P. J. Marks, J. Appl. Cryst., 1994, 27, 200